The Miller group takes a mechanism-guided approach to the discovery of new catalysts for the sustainable synthesis of chemicals and fuels. Our approach starts with the design and synthesis of transition metal catalysts, then shifts to examining catalyst performance with a focus on understanding reaction mechanism in order to inform catalyst improvements.
Collaborative efforts play a major role in the Miller group. The Center for Hybrid Approaches in Solar Energy to Liquid Fuels (CHASE), headquartered at UNC, seeks to develop molecule/material hybrid photoelectrodes for cooperative sunlight-driven generation of liquid fuels from carbon dioxide, nitrogen, and water. A collaborative project targeting electrochemical dinitrogen fixation is supported by the NSF’s Critical Aspects of Sustainability program, with efforts in both chemistry and economics at UNC, Yale, and Rutgers. The Center for Integrated Catalysis is a Phase I NSF Center for Chemical Innovation with groups at UCLA, Boston College, the University of Houston, and UNC. The Miller group also has a tradition of collaborating with chemical industry. This provides researchers in the group the opportunity to interact closely with scientists in chemical industry and to work on projects of direct relevance to commercial chemical processes.
A few ongoing research themes are highlighted here.
Externally Controlled Catalysis: Switchable and Tunable Reactivity
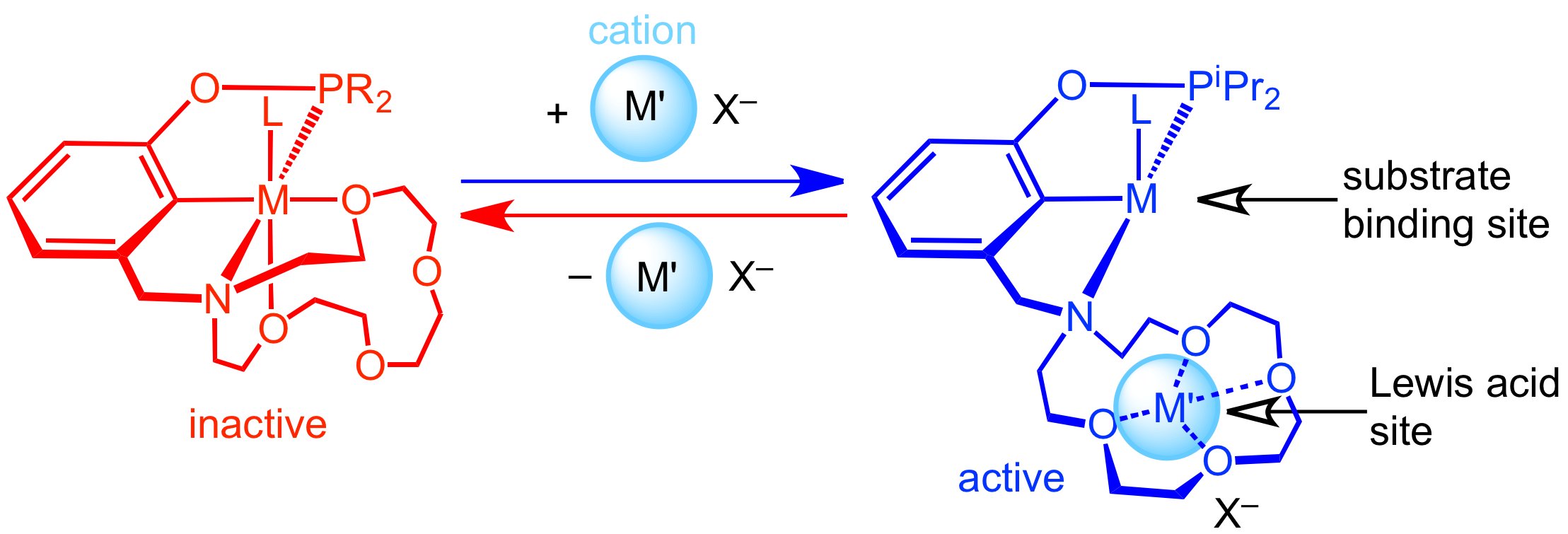
Chemists are constantly seeking new ways to control catalytic activity, selectivity, and longevity. The traditional (and still highly successful) approach to catalyst discovery involves tuning metal and ligand properties until a desired reaction outcome is obtained. An alternative and complementary approach involves preparing a single catalyst that can respond to external additives (also called stimuli or cofactors) to adopt multiple different structures, each with unique reactivity. This “controlled catalysis” approach could streamline catalyst discovery and enable new reactivity traits, such as temporally controlled generation of different products with a single catalyst system or preparation of advanced copolymer morphologies. To explore these applications, organometallic chemists need an improved fundamental understanding of how to design and employ controlled catalysts. We have developed a new family of pincer ligands that utilize an aza-crown ether as one of the donor arms to promote cation-controllable reactivity based on cation-macrocycle interactions occurring near the active site of a transition metal catalyst. The Miller lab is exploring small molecule activation chemistry that takes advantage of the unique pincer-crown ether ligand motif.
Metal Hydride Photochemistry and Thermochemistry
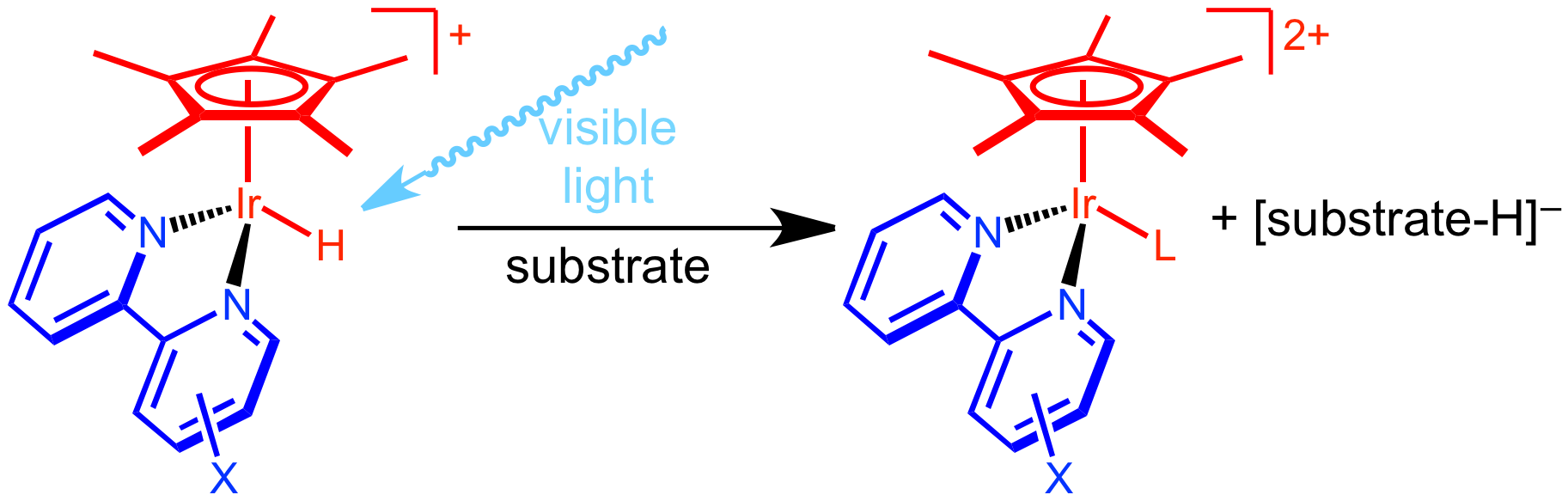
Hydride transfer is a critical step in many reactions, including energy-storing reactions to produce chemical fuels, such as hydrogen evolution and CO2 reduction. What if the hydride transfer step could be controlled using visible light? The ability to trigger a hydride transfer at will, or generate a more reactive species using visible light, could lead to dramatically improved catalysts. We are working to realize this goal by understanding the thermodynamic and kinetic factors associated with excited state hydride transfer to a range of substrates.
Dinitrogen Reduction
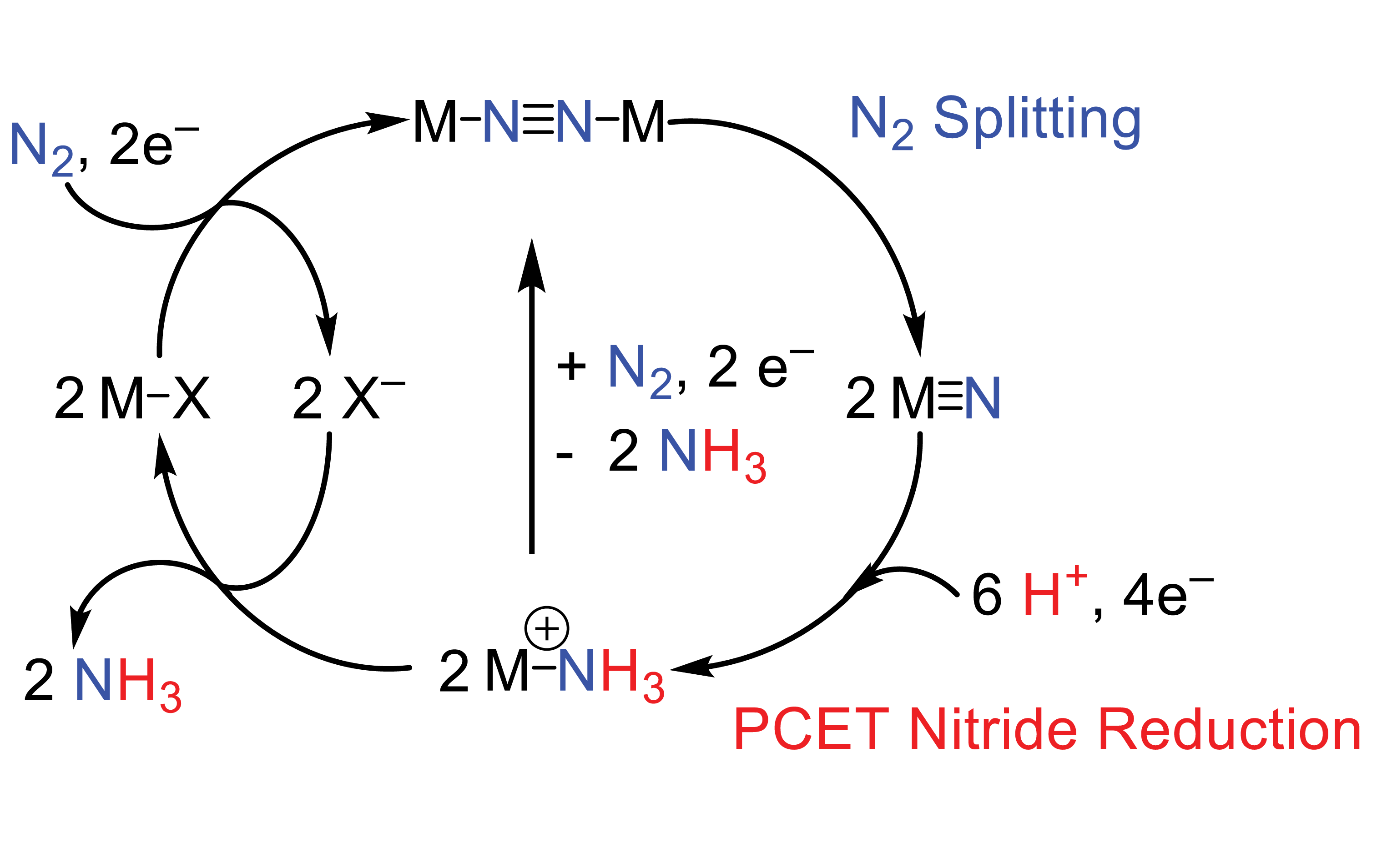
The catalytic synthesis of ammonia via the Haber-Bosch process stands as one of the most transformative chemistry discoveries in history. Because this process relies on fossil-fuel-derived hydrogen gas, however, current ammonia synthesis methods have dire environmental consequences. We envision an alternative ammonia synthesis based on electrocatalytic reduction of N2 to ammonia. Our research in this area is focuses on a pathways in which reductive N2 binding and formation of an end-on bridging N2 complex is followed by splitting into a pair of metal nitrides, followed by proton-coupled electron transfer (PCET) to release ammonia.
Carbon Dioxide Reduction
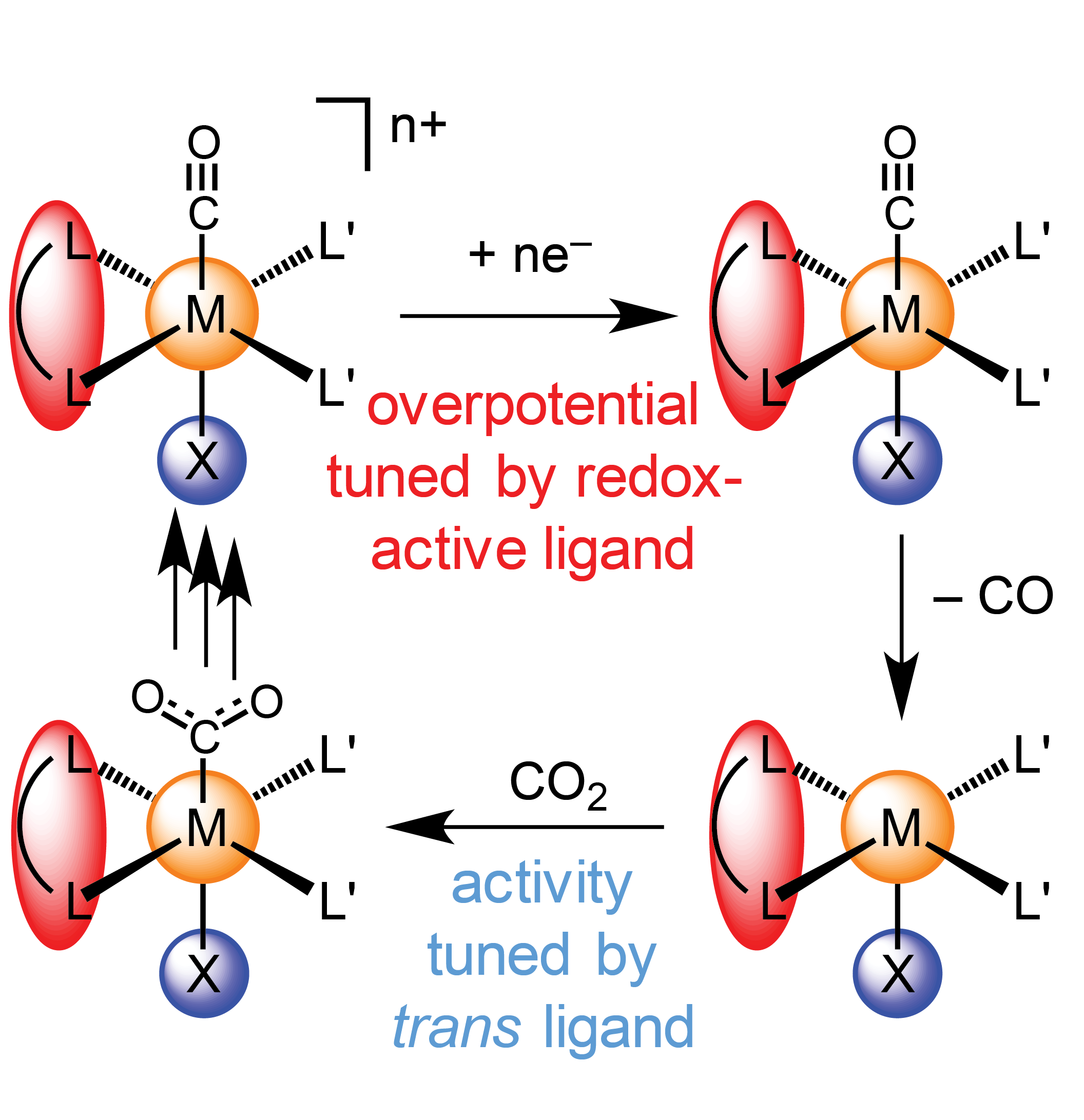
The reduction of carbon dioxide to fuels represents a promising approach to sustainable energy conversion. Molecular electrocatalysts hold promise in this area due to their high selectivity and tunability. We have focused on catalysts that pair one “redox-active” ligand with another strong-trans-effect ligand. By minimizing electronic coupling between the site that accepts electrons and the site that controls the rates of key chemical steps, activity and overpotential can be tuned independently. Although the trans effect is a foundational principle of organometallic chemistry, it had not been systematically studied in the context of CO2 electroreduction.